Shireen Marshall

B.A. Franklin and Marshall College
Ph. D University of Utah (2002)
Magnetic Coordination Networks
The dicyanamide ligand, [N(CN)2]-, was used by former group member Jamie Manson to create materials of composition MII[N(CN)2]2 and MII[N(CN)2]2Lx (L = pyridine, pyrazine, 2,2'-bipyridine, 4,4'-bipyridine).¹ These compounds showed interesting magnetic behaviors with ordering at temperatures as high as 47 K (Cr[N(CN)2]2). In the homoleptic parent compounds, all three nitrogens of the dicyanamide ligand are coordinated and the magnetic exchange between the metals is strong. However, when additional ligands are introduced, dicyanamide is forced to bridge only through the nitrile nitrogens and the magnetic interaction becomes weakly antiferromagnetic.
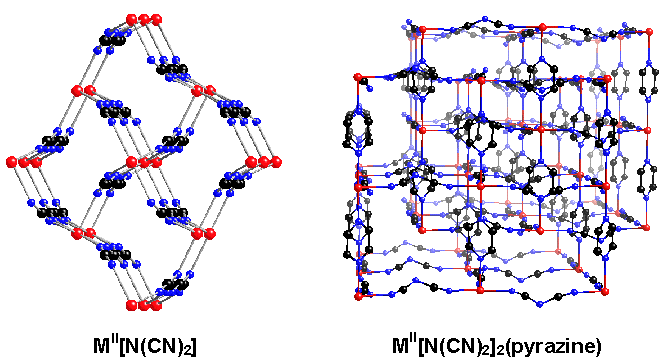
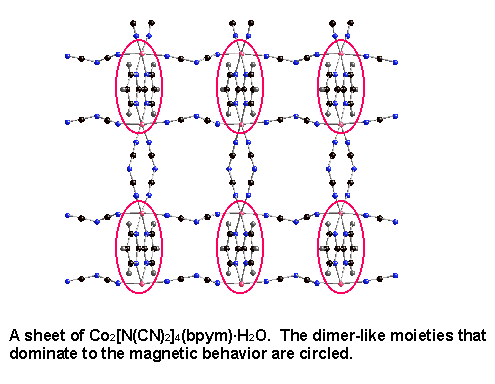
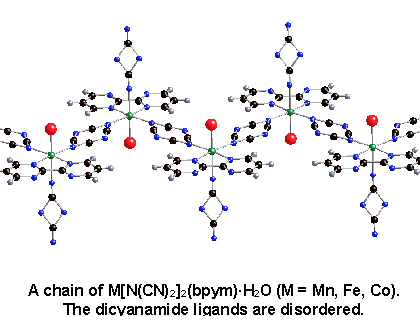
In my work involving the 2,2'-bipyrimidine ligand, we took advantage of the weak coupling of m1,5-bound dicyanamide. The dicyanamide ligand was used to magnetically isolate areas of strong coupling through bis-bidentate bipyrimidine (see structure below). Dimer-like magnetic behavior, apparent as a broad maximum in the magnetic susceptibility data, was observed. This behavior was compared with the very weak antiferromagnetic coupling seen when bipyrimidine was bound only as a bidentate ligand.²
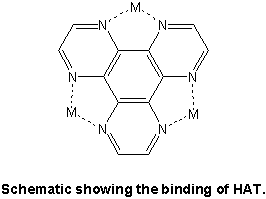
Currently, we are using the ligand 1, 4, 5, 8, 9,12-hexaazatriphenylene (HAT) to try to create similar compounds, which may show trimer-like magnetic behavior.
References
1. (a) Manson, J. L.; Incarvito, C. D.; Rheingold, A. L.; Miller, J. S. J. Chem. Soc., Dalton Trans.1998, 3705. (b) Manson, J. L.; Kmety, C. R.; Huang, Q.; Lynn, J. W.; Bendele, G.; Pagola, S.; Stephens, P. W.; Liable-Sands, L.; Rheingold, A. L.; Epstein, A. J.; Miller, J. S. Chem. Mater.1998, 10, 2552. (c) Manson, J. L.; Miller, J. S. Inorg. Chem.1999, 38, 2552. (d) Manson, J. L.; Incarvito, C. D.; Liable-Sands, L.; Rheingold, A. L.; Miller, J. S. J. Solid State Chem.1999, 145, 369.
2. Marshall, S. R.; Incarvito, C. D.; Manson, J. L.; Rheingold, A. L.; Miller, J. S. Inorg. Chem.2000, 39, 1969.
