Joel S. Miller

INORGANIC, ORGANIC, PHYSICAL & MATERIALS CHEMISTRY
Distinguished Professor
B.S., Wayne State University, 1967
Ph.D., University of California, Los Angeles, 1971
Postdoctoral Associate, Stanford University, 1972
Phone: 801-585-5455
Office: 2124 HEB-N
Email: jsmiller@chem.utah.edu
Research Group
Joel is not recruiting new Ph.D. graduate students into his lab.
Activities & Awards
- American Physical Society's James C. McGroddy Prize for New Materials, 2007
- State of Utah Governors Medal for Science and Technology, 2004
- Utah Award From the Central Utah & Salt Lake Sections of the American Chemical Society, 2004
- Gerhard M.J Scmidt Lecturer, Weizmann Institute of Science, 2003
- Distinguished Research/Creative Award, 2001 University of Utah
- American Chemical Society Award for Chemistry of Materials, 2000
- Japan Society for the Promotion of Science Fellow, 2000
- Wayne State University, Distinguished Alumni Award, 1998
- Manchot Research Professor, Technical University of Münich, 1996
- Visiting Scientist, Weizmann Institute of Science, Rehovot, 1985, 1996
- Industrial Fellow, Northwestern University Material Research Center,1991-93
- Visiting Professor of Chemistry, Universite de Paris-Sud, 1991; University of Pennsylvania, 1988; University of California, Irvine, 1981
- Advisory Boards, Advanced Materials; Chemistry- A European Journal; Inorganic Synthesis Corporation
Recent Publicity

In the dawn of magnets made from molecules, Fernando Palacio, Chem. Commun. 2014.
50, 11437.
Gallery of Journal Covers by the Miller Research Group
Research Interests
 New materials based upon molecular chemistry are the focus of our group's research
activities. We strive toward the preparation and characterization of new materials
with fascinating magnetic, electrical, and optical properties. At present, our major
effort is to prepare magnets based on molecular/ organic/ polymeric chemistry. We
have demonstrated that systems comprising radical cations and anions such as those
with alternating chain structures, may exhibit ferromagnetic coupling and in some
cases are bulk (3-D) ferromagnets. The tetracyanoethylene (TCNE) radical anion salt
of decamethylferrocene is a magnet with a Tc of 4.8 K. Furthermore, on a mole basis they are significantly stronger magnets than
iron metal.
New materials based upon molecular chemistry are the focus of our group's research
activities. We strive toward the preparation and characterization of new materials
with fascinating magnetic, electrical, and optical properties. At present, our major
effort is to prepare magnets based on molecular/ organic/ polymeric chemistry. We
have demonstrated that systems comprising radical cations and anions such as those
with alternating chain structures, may exhibit ferromagnetic coupling and in some
cases are bulk (3-D) ferromagnets. The tetracyanoethylene (TCNE) radical anion salt
of decamethylferrocene is a magnet with a Tc of 4.8 K. Furthermore, on a mole basis they are significantly stronger magnets than
iron metal.
 This led us to react bis(benzene)vanadium with TCNE, and an exciting new magnet with
an unexpected Tc above room temperature was prepared. We have also identified a new class of molecule-based
magnets based on Mn(porphyrin)(TCNE) and exciting new materials with unexpected magnetic
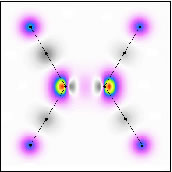
properties are evolving. We are studying the intradimer bonding that occurs for π-[TCNE]22- that has a 2.9 Å intradimer separation, and an unusual 2-electron 4-carbon atom bond.
This bonding arrangement gives rise to an additional CN as well as a new CC vibrations.
Related dimers are being studied.
This led us to react bis(benzene)vanadium with TCNE, and an exciting new magnet with
an unexpected Tc above room temperature was prepared. We have also identified a new class of molecule-based
magnets based on Mn(porphyrin)(TCNE) and exciting new materials with unexpected magnetic
properties are evolving. We are studying the intradimer bonding that occurs for π-[TCNE]22- that has a 2.9 Å intradimer separation, and an unusual 2-electron 4-carbon atom bond.
This bonding arrangement gives rise to an additional CN as well as a new CC vibrations.
Related dimers are being studied.
Our multidisciplinary research targets projects aimed to understand the structure-function relationship to ultimately enable the preparation of stable, technologically useful, high-Tc molecule-based and/or polymeric magnets.
|
|
Film of V[TCNE]x- Room Temperature Sticking to a Magnet. |
|
|
[TCNE]22- Structure with 2e-/4c CC Bond. |
Joel Miller is a member of the Nano Institute of Utah.
Selected Publications
- Thin Film V[TCNE]x Magnets, K.I. Prokhodnya, A.J. Epstein, J.S. Miller, Adv. Mater. 2000 , 12,410.
- Chemical Reduction of 2,4,6-Tricyano-1,3,5-triazine and 1,3,5-Tricyanobenzene. Formation of the 4,4',6,6'-Tetracyano-2,2'-bitriazine and its Radical Anion, R. E. Del Sesto, A. M. Arif, J. J. Novoa, I. Anusiewicz, P. Skurski, J. Simons, B. C. Dunn, E. M. Eyring, J. S. Miller, J. Org. Chem. 2003 , 68, 3367.
- Exceptionally Long ( > 2.9 Å) CC Bonding Interactions in π-[TCNE]22- dimers: Electron - 4 Center Cation-mediated CC Bonding Interactions Involving π* Electrons, R. E. Del Sesto, J. S. Miller, J. J. Novoa, P. Lafuente, Chem. Eur. J. 2002, 8, 4894
- Diruthenium Tetracarboxylate Monocation, [Ru2(O2CR)4]+, Building Blocks for 3-D Molecule-based Magnets, T. E. Vos, Y. Liao, W. W. Shum, J-H. Her, P. W. Stephens, W. M. Reiff, J. S. Miller, J. Am. Chem. Soc. 2004, 126, 11630.
- Enhancement of the Magnetic Ordering Temperature (and Air Stability) of a Mixed Valent Vanadium Hexacyanochromate(III) Magnet to 99 °C (372 K), Ø. Hatlevik, W.E. Buschmann, J. Zhang, J.L. Manson, J.S. Miller, Adv. Mater. 1999, 11, 914.
- Designer Magnets, J.S. Miller, A.J. Epstein, Chem. & Eng. News, 1995, 73 (#40), 30.
- Organic and Organometallic Magnetic Materials Designer Magnets, J.S. Miller, A.J. Epstein, Angew. Chem. Internat. Ed . 1994, 33, 385.
- Cyanil, Synthesis and Characterization of the Strongest Isolated Electron Acceptor and its Reduced Forms, Vazquez, J.C. Calabrese, D.A. Dixon and J.S. Miller, J. Org. Chem. 1993, 58, 65.
- meso-(Tetraphenylporphyrinato)manganese III Tetracyanoethenide [Mn(III) TPP][TCNE]. A Linear Chain Magnet with a Tc of 18 K, J.S. Miller, J.C. Calabrese, R.S. McLean, A.J. Epstein, Adv. Mater. 1992, 4, 498.
- A Room Temperature Molecular/Organic-Based Magnet, J.M. Manriquez, G.T. Yee, R.S. McLean, A.J. Epstein, J.S. Miller, Science 1991, 252, 1415.
- Hexaazaoctadecahydrocoronene, HOC. Structural and Physical Properties of [HOC]n (n = 0, 1+, 2+, 3+, 4+), J.S. Miller, D.A. Dixon, J.C. Calabrese, C. Vazquez, P.J. Krusic, M.D. Ward, E. Wasserman, R.L. Harlow, J. Am. Chem. Soc. 1990, 112, 381.
- Molecular Electronics. J.S. Miller, Adv. Mater. 1990, 2, 378; 495; 601.
- One-Dimensional Inorganic Complexes, J.S. Miller, A.J. Epstein, Prog. Inorg. Chem. 1976, 20, 1.


![[TCNE]22](/_resources/images/faculty/miller_4.jpg)